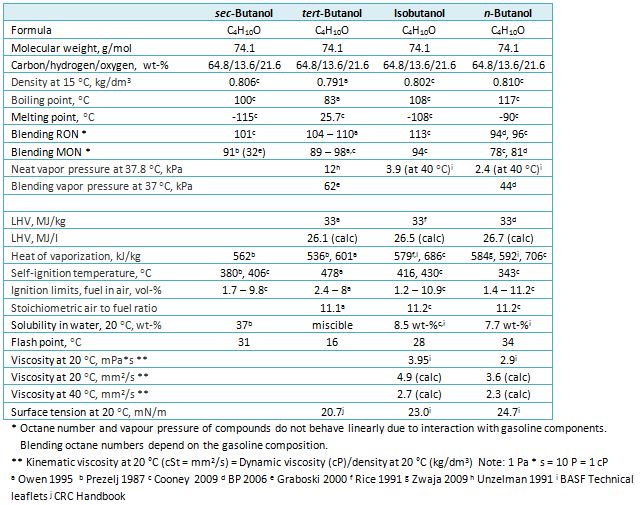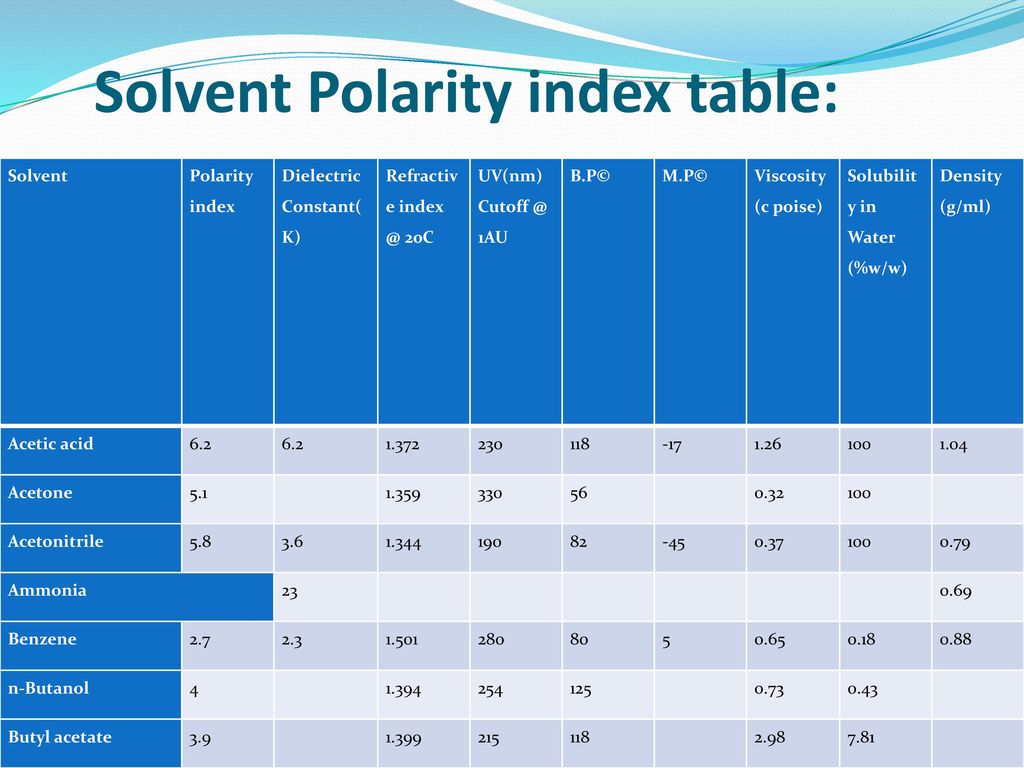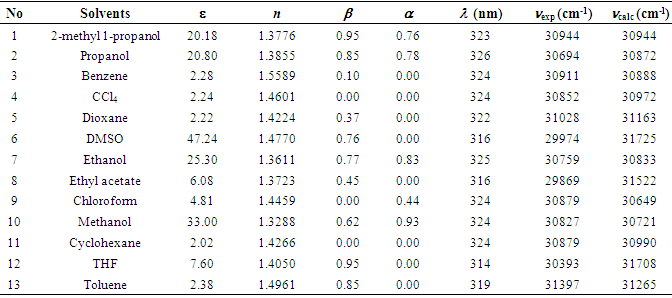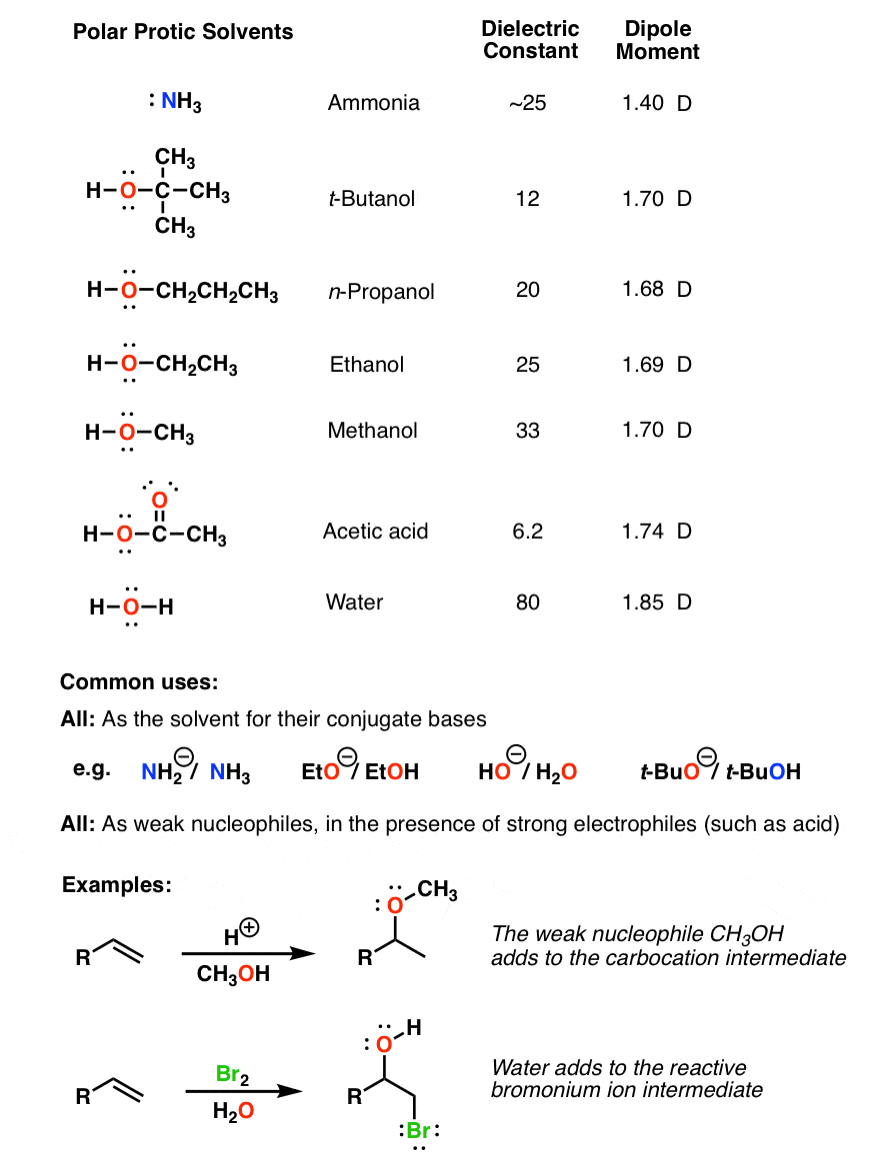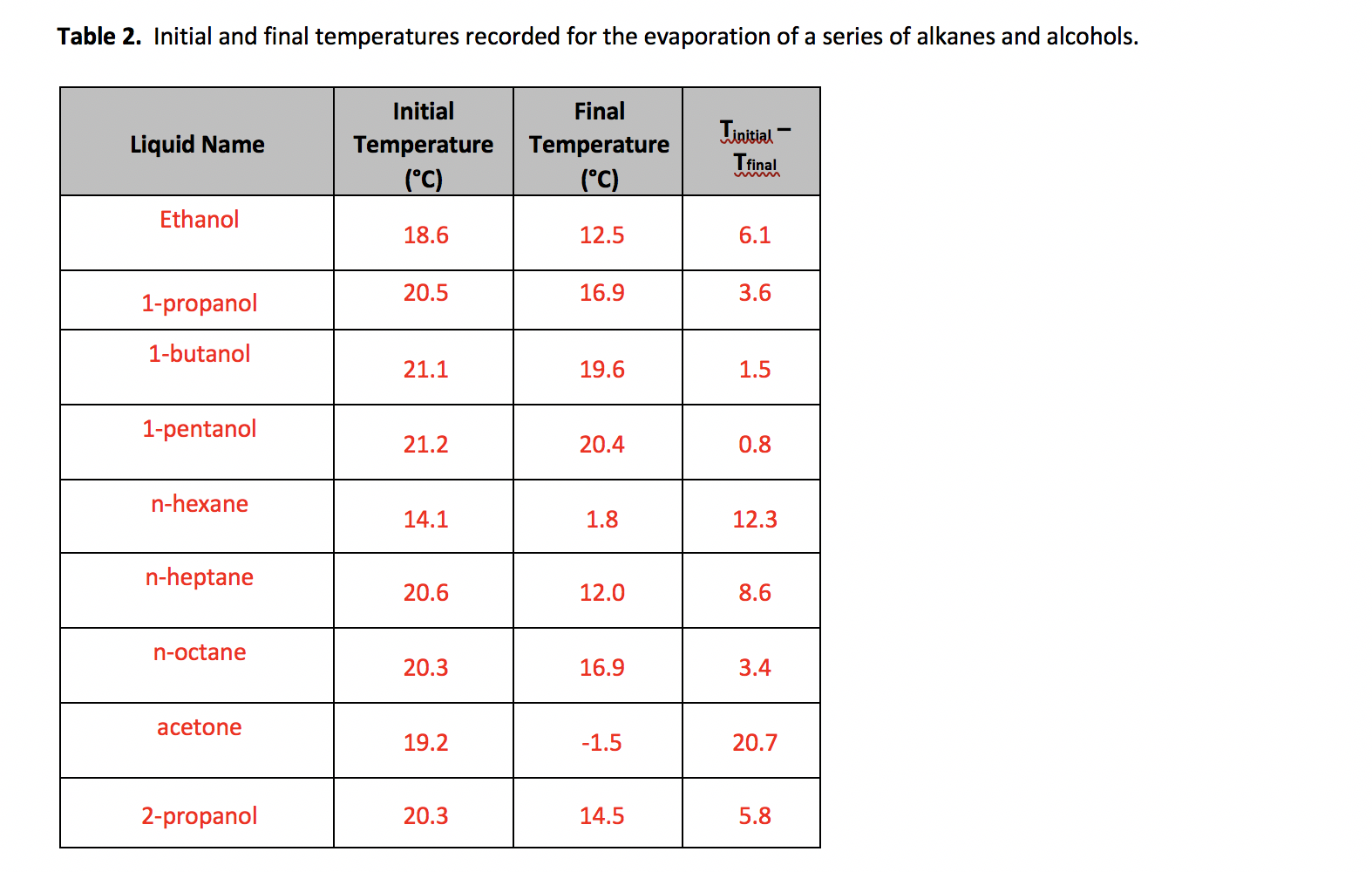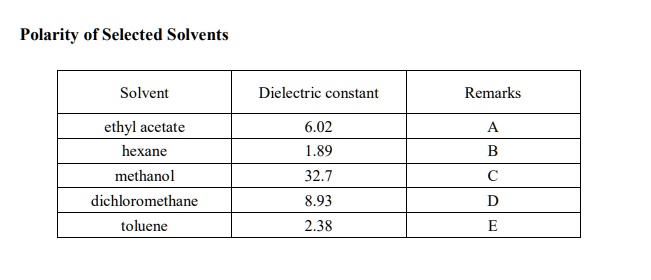
SOLVED: Polarity of Selected Solvents Solvent Dielectric constant Remarks ethyl acetate hexane 6.02 1.89 methanol 32.7 8.93 2.38 dichloromethane toluene
Solubility Measurement and Correlation of Probenecid in 12 Pure Organic Solvents and Thermodynamic Properties of Mixing of Solut
Solvent Polarity and Miscibility How are the relative polarities calcualted? Also, why is methanol less polar than ethanol? (Refer to link highlighted in blue please). - Quora

Preparation of medicinal plants: Basic extraction and fractionation procedures for experimental purposes Abubakar AR, Haque M - J Pharm Bioall Sci

Solvent polarity studies of highly fluorescent laser dye ADS740WS and its fluorescence quenching with silver nanoparticles

n‐Butanol: An Ecologically and Economically Viable Extraction Solvent for Isolating Polar Products from Aqueous Solutions - Reetz - 2021 - European Journal of Organic Chemistry - Wiley Online Library

Table 1 from Solubility of Artemisinin in Different Single and Binary Solvent Mixtures Between (284.15 and 323.15) K and NRTL Interaction Parameters | Semantic Scholar
![FL2 C/P #10] How is "2-butanone" more polar than "n-butanol"? I thought presence hydroxyl makes things more polar? : r/Mcat FL2 C/P #10] How is "2-butanone" more polar than "n-butanol"? I thought presence hydroxyl makes things more polar? : r/Mcat](https://preview.redd.it/dmcr3fgx2pl11.jpg?auto=webp&s=6d5d8ea8d4b68ac45dc2abb75c344cfd1da8515a)